 While arranging the elements according to their atomic weight, if he found that they did not fit into the group he would rearrange them. In 1869, Russian chemist Dmitri Mendeleev created the framework that became the modern periodic table, leaving gaps for elements that were yet to be discovered. He arranged the elements in eight groups but left no gaps for undiscovered elements. He found that every eight elements had similar properties and called this the law of octaves. For example, carbon, being12 times heavier than hydrogen, would have an atomic weight of 12.īritish chemist John Newlands was the first to arrange the elements into a periodic table with increasing order of atomic masses. They concluded that hydrogen would be assigned the atomic weight of 1 and the atomic weight of other elements would be decided by comparison with hydrogen. He arranged them in groups of three in increasing order of atomic weight and called them triads, observing that some properties of the middle element, such as atomic weight and density, approximated the average value of these properties in the other two in each triad.Ī breakthrough came with the publication of a revised list of elements and their atomic masses at the first international conference of chemistry in Karlsruhe, Germany, in 1860. Forty years later, German physicist Johann Wolfang Döbereiner observed similarities in physical and chemical properties of certain elements. 
In 1789, French chemist Antoine Lavoisier tried grouping the elements as metals and nonmetals. As with Chemistry, the text of Wikipedia is available under the GNU Free Documentation License.Among the scientists who worked to created a table of the elements were, from left, Antoine Lavoisier, Johann Wolfang Döbereiner, John Newlands and Henry Moseley. 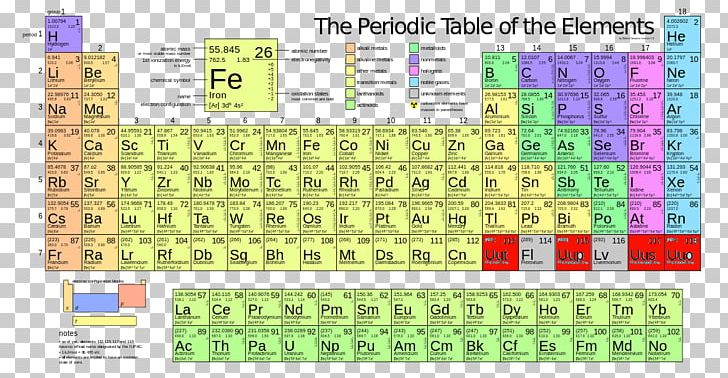
The list of authors can be seen in the page history. The original article was at List of elements by atomic mass. Series: Alkalis - Alkaline earths - Lanthanides - Actinides - Transition metals - Poor metals - Metalloids - Nonmetals - Halogens - Noble gasesīlocks: s-block - p-block - d-block - f-block - g-block Name | Atomic symbol | Atomic number | Boiling point | Melting point | Density | Atomic mass Standard table | Vertical table | Table with names | Names and atomic masses (large) | Names and atomic masses (small) | Names and atomic masses (text only) | Inline F-block | Elements to 218 | Electron configurations | Metals and nonmetals | Table by blocks | Alternatives Atomic weights of elements with atomic numbers 110-116 taken from this source. IUPAC Standard Atomic Weights Revised (2005).Atomic weights of elements with atomic numbers from 1-109 taken from this source. Atomic Weights of the Elements 2001, Pure Appl.Note 5: The atomic weight of commercial Lithium can vary between 6.939 and 6.996-analysis of the specific material is necessary to find a more accurate value.Note 4: The isotopic composition varies in terrestrial material such that a more precise atomic weight can not be given.Note 3: The isotopic composition of the element can vary in commercial materials, which can cause the atomic weight to deviate significantly from the given value.Note 2: The isotopic composition of this element varies in some geological specimens, and the variation may exceed the uncertainty stated in the table.However, three elements, Thorium, Protactinium, and Uranium, have a characteristic terrestrial isotopic composition, and thus their atomic mass given. , indicates the mass number of the longest-lived isotope of the element. Note 1: The element does not have any stable nuclides, and a value in brackets, e.g.For artificial elements the nucleon count of the most stable isotope is listed bracketsĬhemical series of the periodic table Alkali metals The number in parenthesis gives the uncertainty in the "concise notation" dis given in parenthesis next to the least significant digits to which it applies", e.g., 1.00794(7) stands for 1.00794 ± 0.00007. 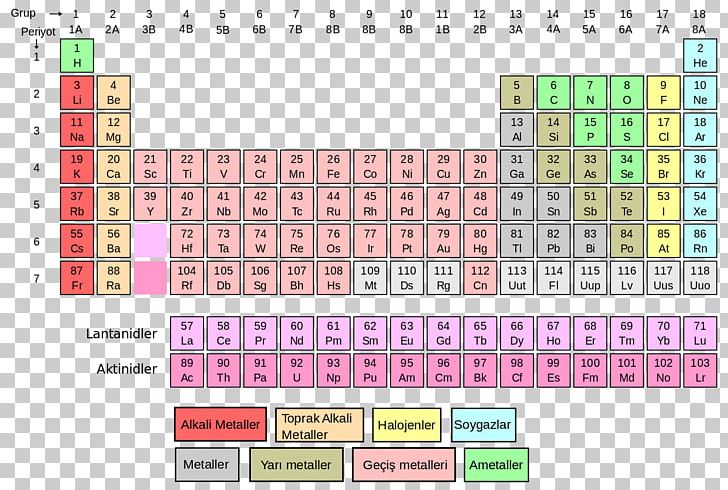
Each element's atomic number, name, element symbol, and group and period numbers on the periodic table are given. This is a list of chemical elements, sorted by atomic mass (or most stable isotope) and color coded according to type of element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
